Diamonds are the best known and are among the most popular stones in the jewellery trade, having been valued for more than 2,000 years. If you have heard about diamonds fluorescing under ultraviolet light but don’t quite know what this means, how it’s relevant to gemmologists, or why it matters to diamond collectors, Pat Daly FGA DGA is here to help. Continue reading to discover more in-depth information about this phenomenon and what causes diamond fluorescence.
Understanding Fluorescence in Diamonds: Why Does it Matter for Gemmologists?
Diamonds are the best known and are among the most popular stones in the jewellery trade and have been valued for more than two thousand years. It was not until the late eighteenth century, however, that diamonds were found to be composed essentially of carbon, and little else about their structure and chemistry was known until the early 20th century. Since then, the increasing industrial importance of diamond has prompted scientists to study it deeply to understand its properties and how they relate to its structure and detailed chemistry.
What are Defects in Diamond?
A notionally perfect diamond would contain only carbon atoms in a regular 3-dimensional structure in which each one was surrounded by four other carbon atoms. Departures from this ideal structure are called defects. Defects, which include chemical impurities and vacancies, are important in gemmology because they may influence the colour of diamonds and luminescent responses, which may be useful in the identification of natural, treated and synthetic stones.
Type 1 vs. Type 2 Diamonds
Diamonds are divided into types according to the quantity and distribution of nitrogen and boron impurities. Type I diamonds contain hundreds of parts per million (ppm) of nitrogen. Type II diamonds contain less than 10ppm (the amount varies somewhat according to the source of information).

[[image: Natural Type 1B diamond producing a bright yellow colour, photographed by Gabriel Kleinberg.]]
Nitrogen in type 1 diamonds is thought to occur as dispersed ions when diamonds are formed. Over the millions of years that diamonds usually reside in Earth’s mantle before they reach the surface, nitrogen atoms come together in pairs and, later, in groups of four surrounding a vacancy, a site from which a carbon atom is missing. Single nitrogen atoms produce a bright yellow colour but little luminescence. The groups of four Nitrogens have no effect on luminescence, and pairs quench it. Neither defect affects colour.
Blue Fluorescence in Long-Wave Ultraviolet
About 98% of natural diamonds contain groups of three nitrogen atoms surrounding a vacancy. This aggregate produces a yellowish colour and blue fluorescence in long-wave ultraviolet, followed by yellow phosphorescence. The amount of these aggregates, balanced by those consisting of pairs of nitrogen ions, determines the luminescence of a stone. It has been reckoned that between a third and all-natural diamonds of this type fluoresce, depending on the ultraviolet wavelength which excites it, the viewing conditions, and the instrument used to observe it. When seen, it is strong evidence that a stone is a natural diamond.

[[image: A natural diamond fluorescing under LWUV (left) and SWUV (right), photographed by Gabriel Kleinberg. ]]
The blue fluorescence is sometimes so strong that it may be noticed when the stone is exposed to the ultraviolet component of daylight. The strength of fluorescence is mentioned on diamond grading reports because some people think it has an adverse effect on the appearance of a stone, although this opinion is not universally shared.
Fluorescence Colours in Diamond
Nitrogen atoms cause visible effects by combining with vacancies in other ways. Two nitrogen ions combined with a vacancy, or four ions with two, cause a green fluorescence. A single nitrogen ion combined with a vacancy causes orange fluorescence.
Yellow fluorescence is thought to result from platelets; planar defects composed mostly of carbon atoms, with some nitrogen.
The colour of pink diamonds is thought to result from dislocations; faults in the atomic structure caused by distortion of the crystal, which may combine with nitrogen. Most natural pink diamonds belong to type I and display blue or yellow fluorescence.

[[image: Diamonds fluorescing under LWUV, photographed by Pat Daly.]]
Fluorescence in Type II Diamonds
Type II diamonds are divided into those with very little nitrogen (type IIa) and a small proportion which contain boron (type IIb). Type IIa stones do not usually show fluorescence with the equipment and in the conditions available to most gemmologists. Some naturally coloured pink diamonds of this type glow orange in long-wave ultraviolet. The cause of this response, and of the colour, is a defect consisting of a nitrogen ion associated with a vacancy. This defect is rare in natural stones; less than one per cent of pink diamonds are coloured by it. It is common, however, in artificially coloured and synthetic pink diamonds.

[[image: The diamonds in a tennis bracelet fluorescing, photographed by Gabriel Kleinberg.]]
Type IIb diamonds contain boron, which is thought to be responsible for the colour of most blue diamonds and their luminescence. Few show any in long wave, but bluish, greenish and orange to red fluorescence is reported in short wave ultraviolet. Phosphorescence after exposure to short-wave is common and may last for a minute. Bluish and greenish are the common colours, but red is well known. It has been found that most natural blue diamonds phosphoresce red, but in many, the effect is masked by the blue-green glow. The balance between the two and the relative rate at which they fade has been proposed as a means of identifying individual stones so that those cut from the same rough or stolen diamonds, which have been recut, may be recognised.
Luminescence in Synthetic Diamond
The luminescence of some synthetic and treated diamonds provides evidence that is easily obtained by gemmologists. Treated pink diamonds, which include synthetic pink diamonds, glow orange in ultraviolet, a rare response in natural pink stones, and high-pressure, high-temperature treated stones, which contain nitrogen, turn to bright yellow and green body colours, and show bright fluorescence in similar colours.

[[image: HPHT synthetic diamond fluorescing under LWUV (left) and SWUV (right), photographed by Gabriel Kleinberg.]]
Blue fluorescence in long-wave ultraviolet is rare in synthetic diamonds. Most of those which fluoresce have an orange to yellow or greenish glow, which is stronger in short wave than in long wave. Many small (melee) diamonds made by HPHT processes contain boron and have a long phosphorescence, which may even be stimulated by daylight. This effect may be diminished by irradiation, but this costs money and may not be done.
Instruments based on the properties of natural and synthetic diamonds in ultraviolet have been made to help the trade screen and identify diamonds. The most sophisticated of these are the Diamond View and SYNTHdetect, made by De Beers.

[[image: CVD synthetic diamond under SWUV (left) and LWUV (right), photographed by Gabriel Kleinberg.]]
Diamond Fluorescence in Short-Wave Ultraviolet
All diamonds fluoresce in very short-wave ultraviolet with a principal wavelength of about 225nm (short-wave lamps most often used by gemmologists produce UV at about 250nm), and the colours emitted by natural and synthetic stones are usually different. Defects accumulate at different rates on different faces of a growing crystal, and they tend to produce different luminescence, so faces present on the growing crystal may be recognised even when it has been polished. Natural stones nearly always grow as octahedra; HPHT synthetic stones have prominent cube faces and CVD-grown synthetics in layers parallel with one pair of cube faces.

[[image: The Aurora Pyramid of Hope Collection of coloured diamonds at the Natural History Museum (left) and the same display under LWUV (right), photographed by Pat Daly.]]
The same kind of short-wave radiation is used in the SYNTHdetect. This instrument is designed so that multi-stone jewellery may be tested. Colours and changes in luminescence over short time periods are used to separate natural from synthetic stones.
The luminescence of diamonds is an interesting and often beautiful feature that may be directly observed and is used in modern instruments to help gemmologists in the difficult tasks of discriminating between natural, treated, and synthetic diamonds.
………………………………………………………………………………………………..
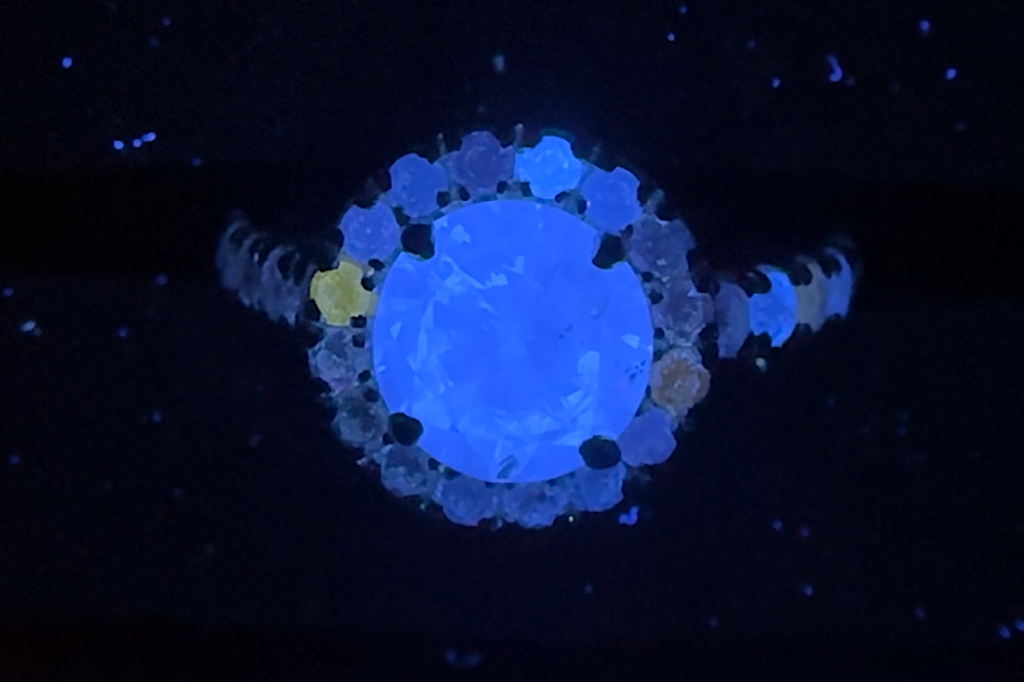
Featured image: Fluorescent diamond ring, photographed by Gabriel Kleinberg.
If you would like to learn more about Gemmology, you can sign up for our short online course GemIntro or explore our accredited programmes.


